HIV Treatments – Looking Ahead in 2020
We’ve slid into 2020, and I’m on holiday, so what better time to look at a summary of the current HIV treatment landscape – what’s available, what’s changed and what’s […]
 Dr Fiona Bisshop
Dr Fiona Bisshop
HIV Doctor,Trans Health Advocate, LGBTI Health Provider, Writer for QNews as "Doc Q"
We’ve slid into 2020, and I’m on holiday, so what better time to look at a summary of the current HIV treatment landscape – what’s available, what’s changed and what’s […]
We’ve slid into 2020, and I’m on holiday, so what better time to look at a summary of the current HIV treatment landscape – what’s available, what’s changed and what’s new and exciting around the corner.
HIV cure is still a long way off, despite what you might read in the newspaper, and new transmissions continue to occur. As more people become aware of the U=U message (undetectable equals untransmittable) antiviral therapy plays a vital role in preventing transmission and reducing stigma.
What’s new in the treatment zone? You could be forgiven for thinking every new HIV treatment announcement these days is simply more of the same – we’ve had good single pill options for a while now, and every new combination pill that comes along feels a bit like an iPhone upgrade rather than something truly better. With each new pill, does this make obsolete the treatment someone has been on for ages and been happy with? Should we be switching to new treatments just because they’re new and supposedly safer/more effective/better tolerated? In some cases, the answer is yes, but reflex switching for the sake of it is hard to justify.
There are new agents coming our way (fostemsavir, islatravir, some monoclonal antibodies, and the new Gilead capsid inhibitor), and new dual therapy options. Long acting injections, are eagerly awaited, and more advanced delivery systems such as implants and micro-array patches are in the wings too.
Looking first at new agents, let’s go to the trusty HIV life cycle to remind ourselves about how these drugs work. All of our current treatments work by interrupting different parts of the HIV replication cycle, starting with attachment of the virus to the CD4 T cell, then the reverse transcriptase enzyme which unzips the viral nucleic acid, making double-stranded DNA, then the integration into the cellular DNA, next viral assembly and capsid formation and finally release into the bloodstream of new viral particles.

Fostemsavir is the first in a new class of treatments called attachment inhibitors (also known as entry inhibitors). By attaching to the gp120 protein on the outer surface of HIV, it blocks the virus from getting into and infecting the CD4 cell. It has no cross-resistance to any other classes of antivirals, so will be useful in people with multi drug resistant virus.
Data from week 96 wk of the BRIGHTE study was presented at IAS last year – the population studied was made up of heavily treated patients failing their current regimen with viral loads >400 and no fully active regimen available – so these were unwell people with no good treatment options.
They split them into 2 groups – those with no active agents left, and those with 1 or more active agents left. Both groups were put onto fostemsavir plus an optimized background regimen (that is, the best combination their clinicians could come up with).
In the group with any remaining active agents, 60% became suppressed by wk 96, and even in the more resistant group, over 40% were suppressed, which is pretty impressive! There was also a modest increase in CD4 count out to 48 wk.
Fostemsavir is likely to be a very useful addition for those patients with limited treatment options, and although there’s currently no pending application for its use in Australia, it is available from ViiV through a compassionate access program.
Onwards now to Islatravir (which is one of the best drug names to appear in recent years in my opinion, sounding like something out of a Tolkien novel and evoking visions of a mighty battle). It was originally known as EFdA – 4 ethinyl 2 fluoro 2 deoxyadenosine, then as MK 8591, so the new name was sorely needed.
Islatravir (ISL) is a 1st-in-class NRTTI (nucleoside reverse transcriptase translocation inhibitor) with multiple mechanisms of action. It has 10-fold greater potency than other approved antiretrovirals, is active at low doses and has a long half-life, allowing for dosing intervals of at least 1 week.
ISL has impressive pharmacodynamics – a single oral dose in naïve subjects led to a greater than 1 log drop in viral load (that’s quite a lot) at doses ranging from 0.5 to 30mg. It has a high barrier to resistance, and is well tolerated with few adverse events reported. There are plans afoot to create a drug-eluting bio-erodable implant for use as PrEP.
In my opinion, the really exciting thing about ISL is that it is going into phase 3 studies paired with doravirine.
So let me remind you about doravirine (DOR) – it’s not a new drug but it’s newly available here in Australia. It’s an NNRTI (non-nucleoside reverse transcriptase inhibitor), and its main selling point is that it’s active against NNRTI-resistant virus, which means it can be used in people with the K103 mutation. There are no baseline cutoffs or limitations in terms of viral load or CD4 count.
It is available alone as Pifeltro (terrible name – I can’t hear it without imagining something rude) and combined with TDF and 3TC as Delstrigo (an OK name, but connotations of Mexican drug cartel overlord).
It’s TGA approved for naïve patients, and Merck will likely apply for approval for switch next year, which is probably where its main market will lie.
It’s hard to see these current formulations containing DOR being widely used here in Australia though. Whilst it has the advantage of being in an STR (single tablet regimen), it’s with TDF rather than TAF. But perhaps the biggest limitation is that we don’t have any data showing a head-to-head comparison with what are now considered first-line – the integrase inhibitor regimens. And like so many new drugs, surprise surprise we really don’t have any safety data about the use of the drug at the time of conception or during pregnancy. So what about the pairing with islatravir?
ISL/DOR (which is simply crying out to be called Isadora) is coming soon to a trial site near you. The phase 2b results published in 2019 showed that this dual therapy was as effective as triple therapy with Delstrigo in maintaining viral suppression. Phase 3 trials will start recruiting early in 2020, and will include a variety of different patient types including those with multiple drug resistant HIV. I’m really looking forward to these studies, which will hopefully validate a robust new therapy option.
Next we have the Gilead capsid inhibitor with the snappy name of GS-6207. Phase 1 data was presented in 2019, showing drug detectable up to 32 weeks after a single SC injection, supporting the amazing possibility of a potential dosing interval of 6 months!! It shows potent antiviral activity and, whilst passaging studies have shown that resistance can develop, it does not lead to resistance towards any other ARV class, and so far there has been no treatment-emergent resistance. There were no serious adverse events and it was generally well tolerated, with injection site reactions being fairly common but resolving rapidly.
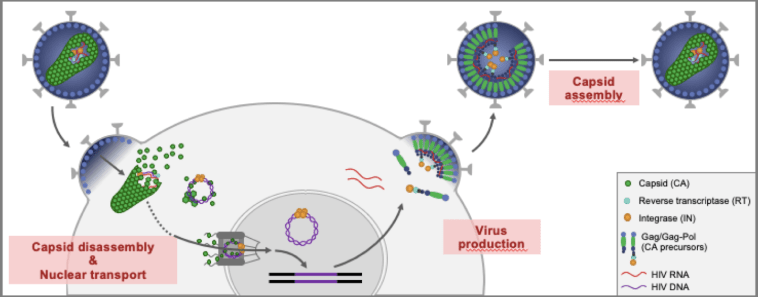
Gilead are moving ahead with phase 2/3 studies next year using GS-6207 with other antivirals, in both naïve and multi drug resistant populations. It’s likely that a capsid inhibitor-based regimen will eventually offer a long-acting SC alternative to the long-acting IM injectables about to be rolled out by ViiV. The key of course will be to find the right dance partner for the capsid inhibitor – hmm I wonder what other long-acting agent would be up for the job?
Ibalizumab is a monoclonal antibody (mAb) which binds to CD4 cells at the gp120 site, and is the first mAb approved by the FDA for HIV treatment in combination with other antivirals in cases of triple ARV resistance. It’s an IV infusion given every 2 weeks.
In heavily treatment experienced patients 40-50% were able to suppress virus, although they were more likely to with a higher baseline CD4. Diarrhoea is the main adverse event, but the big drawback apart from the fact it’s an IV infusion is the cost – so although it remains an option for multi drug resistant HIV, it’s unlikely to be used much.
UB-421 also targets CD4 but at a different site to ibalizumab. Also an IV infusion, it was tested in people suppressed on their current ARVs, and all subjects maintained suppression at <200 copies out to 16 weeks after a single infusion. A lot of people got a rash, and there was some low level viraemia; phase 3 studies are starting next year. This mAb could turn out to be very useful if someone needs a treatment interruption from their usual meds, for instance if starting chemotherapy.
There are plenty of dual therapy studies reported and ongoing, some with new drugs and some with new combinations of old drugs.
The ATLAS 2M study is a phase 3 non-inferiority study comparing long-acting cabotegravir/rilpivirine (CAB/RPV LA) administered as two IM injections every 4 weeks or every 8 weeks over 48 weeks. The study has met its primary endpoint, showing non-inferiority. We have already heard from the earlier ATLAS and FLAIR studies that people really did cope surprisingly well with the monthly injections, and most preferred them to taking daily pills. ATLAS 2M has shown that we can now treat HIV with only 6 injections a year, which is a major advance.
The trade name for the injection is Cabenuva (was someone channelling Barry Mannilow when they came up with this name?). This will be the first completely injectable HIV treatment, so it’s hard to know what it will look like in practice. Obviously injections are not for everyone, but they provide an alternative to daily pills, useful for those who have trouble remembering to take their meds or who simply don’t want the daily reminder of their status. They could also be useful for people planning long periods of travel. We will need good protocols to manage missed injections, and also how to come off a medication which may remain in the system for many many months (the so-called “long tail” of CAB). There are a number of other unanswered questions regarding the injections, and many of these are being addressed in smaller studies.

In a surprise move the US FDA recently rejected an application for Cabenuva, citing concerns about the chemical manufacturing process, however it is likely they will approve it at some stage in 2020. The Australian TGA application for LA CAB/RPV will include the ATLAS 2M data, so when it does finally receive approval it will be for the 2 monthly injection, and will also include oral CAB for the oral lead-in phase when starting this combination for the first time; but we won’t be dancing the Cabenuva here in Australia until at least 2021.
The first fixed dose dual therapy pill Dovato (rhymes with tomato, unless you’re American, in which case I can’t help you) has just been PBS approved here for naïve patients, and there will be an application for switch later this year. All of the major international treatment guidelines (EACS, DHHS, BHIVA) now list Dovato as an option for both switch and initiation under certain circumstances.
It’s hard to gauge exactly how popular this option will be amongst clinicians. Up until now there has been a fair amount of hesitancy largely due to concerns about long term robustness and the risk of resistance. Later this year we will have a lot more data (144 wk of naïve data with GEMINI and 96 wk of switch data with TANGO), so the question is, just how long do these studies have to be before prescribers will feel comfortable initiating with Dovato? Assuming we continue to see no signal for resistance, will that alleviate concerns enough? What’s missing from the data set? Most obviously people with previous failure (although a study is enrolling to look at this population), and there were low numbers of women in the trials as usual, and no pregnancy or breastfeeding data.
Presumably most people would not feel comfortable initiating Dovato in a newly diagnosed person without the results of a baseline resistance assay, meaning the combination would not be suitable for same day initiation or early test and treat. Given the low prevalence of the M184V mutation in Australia, is this really an issue? I think it probably is, especially when we have good treatment alternatives available here.
It’s clear that dual therapy incorporating integrase inhibitors is here to stay. Proposed benefits are that toxicity is minimised, regimens are simpler, and there is the possibility for savings with the availability of generics. There are still some unknowns, and of great interest currently is the question around weight gain with integrase inhibitors, possibly worsened by TAF – this is worthy of its own separate blog! There is a clear signal of weight gain across the class, with DTG being the worst offender, however it appears quite selective, being most evident in black women. There is no clear understanding of why it happens, but an effect on fat cell metabolism has been proposed.

Can you imagine an implant that provides both contraception and HIV prevention (PrEP)? This is under development! And although the main interest is in implants for PrEP, there’s no doubt that implants will also feature in the HIV treatment landscape of the future. The possibility of medication being delivered 24/7 with no thought required from the individual will usher in a new era of reliable HIV treatment.
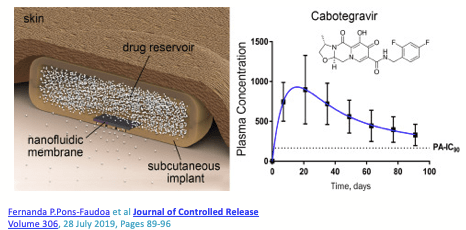

Currently there are studies in healthy volunteers looking at CAB, ISL and TAF implants, and it won’t be long before we see combination ARV implants being developed for use in HIV positive people.
I see a future where patients come in every 12 months for their annual implant replacement….

Finally, exciting new technology is putting nanosuspensions of drugs into microarray patches, which are made up of rows and rows of miniature needles. The patch is applied painlessly to the skin for a matter of seconds, and the weekly dose of medication is delivered just like that! This is still in very early development, but there is the potential here for discreet, long-acting self-administered treatment.
So what will HIV treatment look like in 2020? In the era of U=U, effective antiviral therapy is still the cornerstone of management.
In practice, there will be little change to prescribing choices – the current integrase-based single pill options will continue to be the most popular choice for initiation (despite some concerns about weight gain), but there will be a small amount of uptake of dual therapy.
I’m looking forward to more information about weight gain with integrases, and by the end of the year we may have some early data from the exciting ISL/DOR and capsid inhibitor studies, and a possible date for the arrival of long acting injectable therapy.
Stay tuned for updates and happy new year!
It’s very helpful having a person working in this field provide such clear information, Fiona. Often what I read is second or third hand and then interpreted by a journalist. Thanks for making it understandable for me.
LikeLike